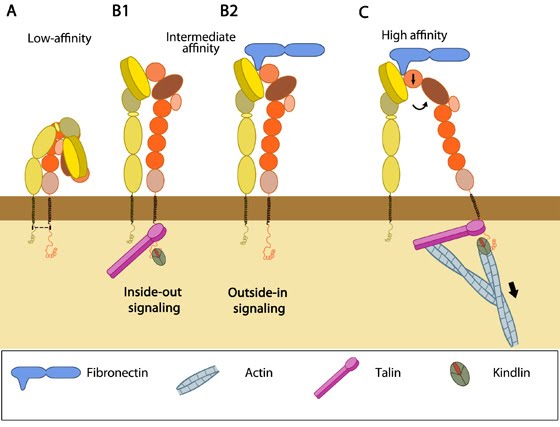
RAP1 GTPase is an important signalling intermediate in integrin activation. Disruption of one of these (the inner membrane clasp) by mutations, or by binding of talin to the β-integrin cytoplasmic domain, can lead to integrin activation. A recent structure of the αIIbβ3 integrin transmembrane domain reveals that the αβ-integrin association is stabilized by two distinct clasps. The activation signal crosses the cell membrane by altering the interaction of the α- and β-integrin transmembrane domains. Interference with these binding events, or lack of these proteins, is associated with genetic defects in integrin activation, and can serve as potential therapeutic targets for various diseases. The final intracellular steps in integrin activation involve the binding of talins and/or kindlins to the β-integrin cytoplasmic domains. Cell-directed changes in the ligand-binding affinity ('activation') of integrins regulate cell adhesion and migration, extracellular matrix assembly and mechanotransduction.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
